What is it necrotizing fasciitis?
Necrotizing fasciitis is very serious. bacterial infection of the Soft fabric and fascia (a sheath of tissue that covers the muscle). the bacteria multiply and release toxins and enzymes that result in thrombosis (coagulation) in the blood vessels. The result is the destruction of soft tissues and fascia
The main types of necrotizing fasciitis are:
- Type I (polymicrobial, that is, more than one bacteria involved)
- Type II (due to hemolytic group A streptococci, staphylococci, including methicillin / MRSA resistant strains)
- Type III (gas gangrene, for example, due to clostridium)
- Other: marine organisms (species of vibrio, Aeromonas hydrophila, considered type III in some reports) and fungal infections (candida and zygomecetes, type IV in some reports).
Necrotizing fasciitis type I
Bacteria that cause type 1 necrotizing fasciitis include Staphylococcus aureus, Haemophilus, Vibrio and several others aerobic and anaerobic strainsEscherichia coli, Bacteroides fragillis) Usually seen in the elderly or in patients with diabetes or other conditions.
Type II necrotizing fasciitis
Type II necrotizing fasciitis has been sensationalized in the media and is commonly referred to as meat-eating disease. It affects all age groups. Healthy people are also prone to infection with this group.
Type III necrotizing fasciitis
Type III necrotizing fasciitis is caused by Clostridia perfringens or less commonly Clostridia septicum. It usually follows a major injury or surgery and produces gas under the skin - this produces a crackling sound called crepitation. Intravenous drug users who inject “black tar” heroin subcutaneously can also become infected with clostridia and develop necrotizing fasciitis.
Other organisms
Necrotizing fasciitis due to marine organisms is usually due to contamination of wounds by seawater, cuts from fins or fish stingers, or the consumption of raw shellfish. It occurs most commonly in patients with liver disorders. These infections can be very serious and can be fatal if they are not treated within 48 hours.
Fungal necrotizing fasciitis gets complicated traumatic wound on immunocompromised people.
Other terms used for necrotizing fasciitis include hemolytic streptococcal gangrene, Meleney ulcer, acute dermal gangrene, hospital gangrene, suppurative synergistic fasciitis and necrotizing cellulitis.
Necrotizing fasciitis affecting perinealgenitals and perianal regions is known as Fournier's gangrene. This has a particularly high mortality rate ranging from 15% to 50%.
How do you get necrotizing fasciitis?
Necrotizing fasciitis can occur in anyone - in fact, nearly half of all known cases of strep necrotizing fasciitis have occurred in young, previously healthy individuals. The disease can occur if the correct set of conditions is present, these include:
- An opening in the skin that allows bacteria to enter the body. This can occur after a minor injury (eg, small cut, grazing, puncture, injection) or a large injury due to trauma or surgery (for example, laparoscopy, sclerotherapy, endoscopic gastrostomy, thoracostomy, caesarean section, hysterectomy). Sometimes no entry point can be found.
- Cervicofacial necrotizing fasciitis can follow a jaw fracture or dental infection.
- Direct contact with a person who carries the bacteria or the bacteria is already present in another part of the person.
- Particularly invader strains of bacteria, for example streptococci that evade the immune system and produce a toxin called cysteine protease SpeB, which dissolves tissue.
- In children, type II necrotizing fasciitis can complicate chickenpox. Other causes of necrotizing fasciitis in children include omphalitis, necrotizing enterocolitis, and urachal abnormalities.
The risks of necrotizing fasciitis include:
- Aspirin and nonsteroidal anti-steroidsinflammatory drugs
- Advanced age
- Diabetes
- Immunosuppression
- Obesity
- Drugs abuse
- Serious chronic disease
- Malignancy.
Pathophysiology necrotizing fasciitis
The infection begins in the superficial fascia. The enzymes and proteins released by the responsible organisms cause necrosis of fascial layers. The horizontal spread of the infection may not be clinically apparent on the skin surface and therefore the diagnosis may be delayed. The infection then spreads vertically up the skin and down into deeper structures. Thrombosis occludes the arteries and veins that lead to ischemia and tissue necrosis.
Streptococci produce:
- M proteins, which initiate an inflammatory response with the release of numerous cytokines (L-1, IL-6, TNFα)
- Exotoxins, which destroy neutrophils allowing bacterial growth and destroying tissues.
Aerobic and anaerobic bacteria produce hydrogen, nitrogen, hydrogen sulfide, and methane gases that destroy hyaluronic acid allowing the infection to spread.
What are the signs and symptoms of necrotizing fasciitis?
Signs and symptoms vary between individuals, but some or all of the following are often present.
Initial symptoms
- The most common site of infection is the lower leg. Necrotizing fasciitis can also affect the upper limb, perineum, buttocks, trunk, head and neck.
- Symptoms generally appear within 24 hours of a minor injury.
- The pain is usually very severe on presentation and worsens over time.
- There may be flu-like symptoms such as nausea, fever, diarrhea, dizziness and in general discomfort.
- Intense thirst develops as the body becomes dehydrated.
Symptoms after 3 to 4 days.
As necrotizing fasciitis takes hold:
- The affected area begins to swell and may show a purple color. eruption
- Large dark marks form that turn into blisters filled with dark fluid.
- The wound begins to die and the area turns black (necrosis)
- Edema It is common
- A fine sensation of crackling under the skin (crepitus) is due to gas in the tissues.
- Severe pain continues until necrosis / gangrene destroys peripheral nerves when the pain goes away
- The infection may not improve when antibiotics are given.
Around days 4–5, the patient is very ill with dangerously low blood pressure and high temperature. The infection has spread to the bloodstream and the body enters toxic shock. The patient may have altered levels of consciousness or become totally unconscious.
Metastatic Abscesses can develop in the liver, lung, spleen, brain, pericardium and, rarely, on the skin.
Necrotizing fasciitis
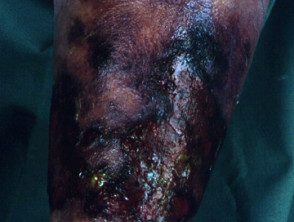
Necrotizing fasciitis
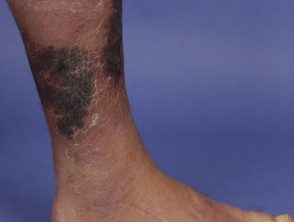
Necrotizing fasciitis
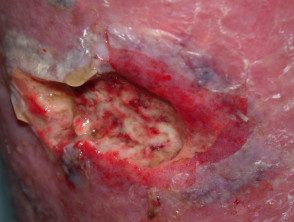
Necrotizing fasciitis
See more images of necrotizing fasciitis ...
How is necrotizing fasciitis diagnosed?
A complete medical history and clinical examination are crucial in reaching the diagnosis of necrotizing fasciitis. Special care should be taken when examining immunocompromised patients, as the presentation of symptoms / signs may be atypical.
- Clostridial and streptococcal infections as a result of a traumatic or surgical wound usually manifest rapidly compared to necrotizing fasciitis due to other organisms.
- A positive finger test is highly pathognomic for necrotizing fasciitis. A vertical of 2 cm. incision it is done on the affected skin and an index finger is pushed into the tissue. The test is positive if the finger passes through the subcutaneous fabric without resistance.
- There is little adhesion of the tissue to the fascia when incising the site.
- Necrotic tissue / pus comes out of the fascial planes.
- The liquid the color of dish water seeps from the skin.
- Necrotizing fasciitis usually does not bleed.
Laboratory screening investigations
These generally show:
- White blood cell count> 15.4 x 109 / L
- Serum sodium <135 mmoL
- High CRP (> 16 mg / dL)
- High CK level (> 600 U / L)
- Urea> 18 mg / dL.
Blood culture, deep tissue biopsy and Gram help identify the culprit organism(s) and guide the choice of antibiotic. If Staphylococcus aureus is detected, a MRSA sensitivity test should be performed. Blood cultures are usually negative for clostridial species.
Fungal culture should be performed in immunocompromised and trauma patients.
Image
Bone scan, Connecticut and Magnetic resonance identify fluid collection areas, inflammation and gas within soft tissues.
Laboratory risk indicator for necrotizing fasciitis
The Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) is a tool that helps distinguish necrotizing fasciitis from other tissue infections based on six parameters. A score of ≥ 6 favors necrotizing fasciitis. This test is not appropriate for all cases and is not completely reliable.
LRINEC parameters
CRP (mg / L) ≥150:
- 4 points
White blood cell count (× 103/ mm3):
- <15: 0 puntos
- 15-25: 1 point
- > 25: 2 points
Hemoglobin (g / dL):
- > 13.5: 0 points
- 11-13.5: 1 point
- <11: 2 puntos
Sodium (mmol / L) <135:
- 2 points
Creatinine (umol / L)> 141:
- 2 points
Glucose (mmol / L)> 10:
- 1 point
What is the treatment for necrotizing fasciitis?
Once the diagnosis of necrotizing fasciitis is confirmed, treatment should begin without delay.
- The patient must be hospitalized and is often admitted to an intensive care unit.
- The causative organism (s) should be identified and treated with high-dose intravenous antibiotics. The initial choice of antibiotics includes penicillin, clindamycin, metronidazole, cephalosporins, carbapenems, vancomycin, and linezolid. After reporting the culture, the choice is adjusted.
- It is absolutely vital that an experienced surgeon urgently remove all necrotic tissue (debridement)
- Supplemental oxygen, fluids, and medications may be needed to raise blood pressure.
- Hyperbaric and intravenous oxygen immunoglobulin It can also be considered.
Immediate surgical debridement improves survival and avoids the complications of necrotizing fasciitis. All infected tissue must be removed using an adequate amount. excision. Repeated debrides are carried out over a few days.
When the acute infection has subsided, the wound should be closed with a skin graft if necessary. Vacuum-assisted wound closure devices can be helpful in healing a persistent ulcer.
What is the likely outcome?
Prompt diagnosis and treatment are essential to reduce the risk of death and disfigurement from necrotizing fasciitis.
If diagnosed and treated early, most patients will survive necrotizing fasciitis with minimal scarring. If there is significant loss of tissue, a subsequent skin graft will be necessary and in some patients amputation of the extremities is required to avoid death.
Up to 25% of patients will die of necrotizing fasciitis, due to complications such as renal failure and septicemia (blood poisoning) and multi-organ failure.

