What is psoriasis?
Psoriasis is a autoimmune skin disorder characterized by circumscribed, Red, scaly plates.
Calcipotriol/Betamethasone Diproprionate Suitable Psoriasis
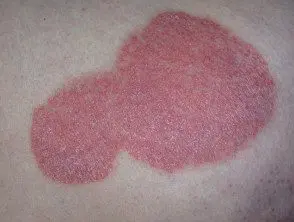
Chronic plaque psoriasis
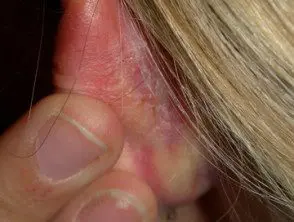
Psoriasis of the scalp.
What is Calcipotriol Dipropionate / Betamethasone? ointment and gel?
Calcipotriol / betamethasone dipropionate gel and ointment are current medications to treat license plate psoriasis in adult patients older than 18 years [1,2]. Brand names for calcipotriol/betamethasone dipropionate are Daivobet® Ointment 50/500 and Daivobet® 50/500 gel. The New Zealand Marketing Authorization Holder is LEO Pharma. Calcipotriol is also called calcipotriene. In some countries, the brand name of the combined product is Dovobet®.
Daivobet® ointment and Daivobet® the gel is available in New Zealand by prescription and is fully funded by PHARMAC.
How does calcipotriol/betamethasone dipropionate work?
The mechanism of action of the ingredients has been studied independently, as well as in combination. [1–3].
Calcipotriol
Calcipotriol hydrate is a synthetic vitamin D analog term.
- In psoriasis, vitamin D and its analogs act primarily for inhibit keratinocytes proliferation and induce keratinocytes differentiation.
- Calcipotriol works by reducing the number of new skin cells that are made and by causing the new cells to mature more quickly.
- the underlying antiproliferative mechanism of vitamin D in keratinocytes involves the induction of transforming growth inhibitory factor growth factor-β and cyclin-dependent kinase inhibitors, with subsequent growth arrest in the G1 phase of the cell cycle.
- Vitamin D also has a immunomodulator effect, suppressing the activation and differentiation of Th17/Th1 cells and inducing a Th2 response.
betamethasone dipropionate
Betamethasone dipropionate is a potent synthetic topical corticosteroid.
- In psoriasis, corticosteroids suppress the immune system, particularlyinflammatory cytokines and chemokines, thus inhibiting T cell activation.
- Betamethasone dipropionate works in psoriasis by reducing inflammation to relieve skin symptoms such as burning, pain, itching and swelling.
The combination of calcipotriol and betamethasone dipropionate
Betamethasone dipropionate and calcipotriol ointment and gel combine the pharmacological effects of betamethasone dipropionate and calcipotriol. The combination of the two drugs has a specially designed formulation that works to both reduce inflammation and treat pain. climbing associated with psoriasis.
- The calcipotriol in calcipotriol/betamethasone dipropionate ointment and gel treats overactive skin cells.
- Betamethasone Dipropionate relieves redness, swelling, itching, and skin irritation.
- The combined treatment flattens psoriasis plaques, removes scaling and reduces discomfort.
- In combination, calcipotriol and betamethasone dipropionate promote greater anti-inflammatory and antiproliferative effects than either component alone.
How is calcipotriol/betamethasone dipropionate administered?
Each gram of calcipotriol/betamethasone dipropionate ointment and gel contains 50 mcg of calcipotriol and 500 mcg of betamethasone dipropionate [1–3].
Adults
Calcipotriol/betamethasone dipropionate ointment and gel are applied topically to affected areas once daily. The recommended treatment period is 4 weeks.
After this period, calcipotriol/betamethasone dipropionate ointment can be used as needed under medical supervision. [1,2]. The maximum daily and weekly doses should not exceed 15 g and 100 g, respectively.
For the application of calcipotriol / betamethasone dipropionate gel, an amount between 1 g and 4 g per day is sufficient for the treatment of the scalp (4 g corresponds to a teaspoon). For optimal effect, it is recommended that hair and the affected skin areas are not washed immediately after the application of calcipotriol/betamethasone dipropionate gel. Calcipotriol/Betamethasone Dipropionate Gel should be left on the affected area overnight or during the day. The gel can also be used for psoriasis on the trunk and extremities.
Children
Calcipotriol/betamethasone dipropionate ointment and gel are not recommended for use in children and adolescents under 18 years of age due to a lack of data on safety and effectiveness [1–3].
Caution
Calcipotriol/Betamethasone Dipropionate Ointment and Gel should not be used:
- With occlusive dressings, unless directed by a doctor
- On the face, groin or armpitsor if the skin atrophy is present at the treatment site
- For oral, ophthalmic or intravaginal use.
Treated skin areas should be protected from sunlight and ultraviolet rays (UV) radiation, for example by wearing sun-protective clothing, since sunlight inactivates calcipotriol.
Additional topical corticosteroids should not be used on areas treated with calcipotriol/betamethasone dipropionate ointment or gel [1–3].
Missing dose of calcipotriol/betamethasone dipropionate ointment or gel
If you miss a dose of calcipotriol/betamethasone dipropionate ointment or gel, apply it as soon as you remember. If it is time for the next dose, the missed dose is skipped. Do not apply a double dose.
How long should calcipotriol/betamethasone dipropionate ointment and gel be used?
Calcipotriol/Betamethasone Dipropionate Ointment and Gel is usually used for 4 weeks, but can be used for up to eight weeks on medical advice. Your doctor may recommend repeated treatments depending on the response to the first treatment.
Which are the contraindications for the use of calcipotriol / betamethasone dipropionate ointment and gel?
The main contraindication a calcipotriol/betamethasone dipropionate ointment and gel is hypersensitivity the active substance or any of the excipients [1–3].
It should not be used for acute types of psoriasis like generalized pustular psoriasis or erythrodermic psoriasis.
Calcipotriol/betamethasone dipropionate ointment and gel should also not be used for skin affected with:
-
viral, fungal, bacterialor parasitic skin infections
- Tuberculosis (TB)
- Newspaper dermatitis
- Ichthyosis
- Acne
-
Rosacea
Before using Calcipotriol/Betamethasone Dipropionate Ointment or Gel, patients should inform treating physicians of the following circumstances:
- if they have calcium metabolism disorder
- If they are receiving phototherapy.
- If they are pregnant or plan to become pregnant (see Safety of Medications Taken During Pregnancy)
- If they are breastfeeding or plan to breastfeed (see Breastfeeding and the skin).
What are the warnings and precautions for using calcipotriol/betamethasone dipropionate ointment and gel?
Hypercalcemia and hypercalciuria
Hypercalcaemia and hypercalciuria have been observed with the use of calcipotriol/betamethasone dipropionate ointment and are more likely if recommended doses (maximum daily dose 15 g; maximum weekly dose 100 g) are exceeded [1–3].
In view of the risk of hypercalcaemia secondary to excessive absorption of calcipotriol when there is extensive skin involvement, calcipotriol/betamethasone dipropionate ointment and gel should not be used on more than 30% of the body surface.
If hypercalcaemia or hypercalciuria develops, treatment should be withheld until calcium metabolism parameters have normalized.
Serum calcium and renal function should be monitored at 3-month intervals during periods of calcipotriol/betamethasone dipropionate ointment or gel use.
effects on the endocrine system
Systemic absorption of a topical corticosteroid, such as betamethasone dipropionate, can rarely produce reversible hypothalamicsuppression of the pituitary-adrenal (HPA) axis with the potential for glucocorticosteroid insufficiency during treatment or when withdrawing the topical corticosteroid.
Factors that predispose a suppression of the axis of the patient with HPA includes:
- Application under occlusive dressings
- Application on large areas of damaged skin.
- Mucous membranes or skin folds that increase systemic absorption of corticosteroids [1–3].
Systemic effects of topical corticosteroids may also include Cushing's syndrome, hyperglycemiaand glycosuria. Local side effects of topical corticosteroids include:
- Cutaneous atrophy
- Stretch marks
- Telangiectasia
- Dryness
- perioral dermatitis
- Secondary infection
-
Miliaria
Use not evaluated
There is no published experience with the use of calcipotriol/betamethasone dipropionate ointment and gel on the scalp or in guttate psoriasis.
Adverse reactions to excipients
Calcipotriol/betamethasone dipropionate ointment and gel contain butylated hydroxytoluene (E321), a excipient within the excipient polyoxypropylene stearyl ether, which may cause local skin reactions (eg, contact dermatitis) or eye irritation and mucous membranes [1,2].
visual disturbance
Visual disturbance has been reported with the use of topical and systemic corticosteroids. If a patient presents with symptoms such as blurred vision or other visual disturbances, consideration should be given to referring the patient to a ophthalmologist for evaluation of possible causes which may include waterfall, glaucomaor rare diseases such as central serous chorioretinopathy, which have been reported after the use of systemic and topical corticosteroids [1–3].
UV exposure
the phototoxic The effects of calcipotriol/betamethasone dipropionate ointment and gel have not been systematically studied in psoriasis patients undergoing phototherapy or exposed to sunlight. Treated skin areas should be protected from sunlight and UV radiation (using sun protective clothing or sunscreens), particularly where exposure may be considerable for reasons such as occupation. [1–3].
Use of calcipotriol/betamethasone dipropionate ointment and gel in specific populations
Children
The safety and efficacy of calcipotriol/betamethasone dipropionate ointment and gel in children under 18 years of age have not yet been established in formal trials [1,2].
Due to a higher skin surface to body mass ratio, children younger than 12 years are at particular risk of systemic adverse effects when treated with topical corticosteroids, including growth retardation [1–3].
Pregnant women
There are no adequate and well-controlled studies in pregnant women. [1,2].
Calcipotriol/betamethasone dipropionate ointment and gel should only be used during pregnancy if the potential benefit to the patient justifies the potential risk to the fetus.
Animal reproduction studies have not been conducted with calcipotriol/betamethasone dipropionate ointment or gel. However, calcipotriol has been shown to be fetotoxic and betamethasone dipropionate has been shown to be teratogenic in animals when administered systemically.
Nursing mothers
Betamethasone is excreted in breast milk. It is unknown whether topical application of calcipotriol/betamethasone dipropionate ointment or gel could result in sufficient systemic absorption to produce significant quantities of this corticosteroid in human breast milk. There are no data on the excretion of calcipotriol in breast milk. [1,2].
Application of calcipotriol/betamethasone dipropionate ointment to the breast area should be avoided to prevent possible ingestion by infants. Calcipotriol/betamethasone dipropionate ointment and gel should only be used during lactation if the potential benefits clearly outweigh the potential risks.
After applying calcipotriol/betamethasone dipropionate ointment or gel to the skin, mothers should wash their hands thoroughly before handling their baby.
Patients with kidney failure.
Safety in patients with renal insufficiency has not been established. Calcipotriol/betamethasone dipropionate ointment and gel are contraindicated in patients with severe renal insufficiency [1–3].
Patients with hepatic disability
Safety in patients with hepatic impairment has not been established. Betamethasone dipropionate/calcipotriol ointment and gel are contraindicated in patients with severe hepatic impairment [1–3].
Patients of childbearing age.
The possible effects of betamethasone in combination with calcipotriol on fertility have not been investigated in animals. Studies of oral administration of calcipotriol in rats have shown no impairment of male and female fertility. [1,2].
What are the potential drug interactions of calcipotriol/betamethasone dipropionate ointment and gel?
No interaction studies have been performed with calcipotriol/betamethasone dipropionate ointment and gel. [1,2]. no experience with concurrent utilizar con otra terapia sistémica para la psoriasis o con fototerapia.
El ungüento de dipropionato de calcipotriol / betametasona no debe usarse simultáneamente con suplementos de calcio o vitamina D, o con medicamentos que mejoren la disponibilidad sistémica de calcio.
What are the possible side effects of calcipotriol/betamethasone dipropionate ointment and gel?
El programa de ensayos clínicos para ungüento y gel de dipropionato de calcipotriol / betametasona ha incluido hasta ahora a más de 2.500 pacientes y ha demostrado que se puede esperar que aproximadamente el 10% de los pacientes experimenten un efecto adverso no grave [1,2].
El único común (<1>adverse event observado en receptores tratados fue pruritus and skin exfoliation.
Los efectos adversos en <1>
- Sensación de ardor en la piel.
- Dolor o irritación de la piel
- Dermatitis
- Erythema
- Atrofia de la piel
- Exacerbation psoriasis
- Eruption
- Erupción pustulosa
- Irritación de ojo.
Because clinical trials are conducted under highly variable conditions, adverse reaction The rates observed in clinical trials of one drug cannot be directly compared with the rates in clinical trials of another drug and may not reflect the rates observed in practice.
Adverse effects associated with the pharmacological class.
Calcipotriol
Las posibles reacciones adversas del calcipotriol incluyen reacciones en el sitio de aplicación, tales como:
- Pruritus
- Irritación de la piel
- Una sensación de ardor o escozor
- Dry Skin
- Erythema
- Eruption
- Dermatitis/eczema
- Psoriasis agravada
- Photosensitivity y reacciones de hipersensibilidad, incluidos casos muy raros de angioedema and facial edema.
betamethasone dipropionate
Pueden ocurrir reacciones locales después del uso tópico de diproprionato de betametasona, especialmente durante la aplicación prolongada, que incluye:
- Atrofia de la piel
- Telangiectasia
- Stretch marks
- Folliculitis
- Hypertrichosis
- perioral dermatitis
- Allergic contact dermatitis
- Depigmentation
- Colloid milia.
Las reacciones sistémicas debidas al uso prolongado de corticosteroides tópicos en grandes áreas de la piel incluyen:
- Supresión adrenocortical
- hypercalcemia
- waterfalls
- Infections
- Impacto sobre el metabolismo control de diabetes mellitus y aumento de la presión intraocular.
Se aconseja a los profesionales de la salud que informen todas las sospechas de reacciones adversas al Centro de Farmacovigilancia de Nueva Zelanda.

